Latest News
14.09.2021
Almus Launch Erectile Dysfunction Relief 50mg Film Coated Tablets into UK Pharmacy Market
According to the British Association of Urological Surgeons (BAUS) erectile dysfunction becomes increasingly more common with age and is seen in 50 - 55% of men between 40 and 70 years old. It is often associated with obesity, high blood pressure, high cholesterol and diabetes, which are all significant risks to overall health*
Almus are pleased to announce the launch of Almus Erectile Dysfunction Relief 50mg Film Coated Tablets to the UK pharmaceutical market.
Tara Dew, Own Brand Manager, said: “We believe that erectile dysfunction is no longer an off-limits discussion. We feel that sufferers should not feel embarrassed to ask for advice from their pharmacists. The launch of Almus Erectile Dysfunction Relief 50mg Film Coated Tablets brings an affordable treatment option to both pharmacists and patients, compared to the UK leading brand competitor.”
Sachiv Patel, Director of National Accounts & Health and Beauty at Alliance Healthcare, comments “This is a landmark product launch for us as we firmly believe that essential patient and personal care products should be available to everyone at an affordable price, without compromising on quality.”
Almus Erectile Dysfunction Relief 50mg Film Coated Tablets will be the first of several new products to be launched in innovative new packaging.
“We’re preparing to launch a range of new lines across various therapeutic categories before the end of the year. and the new packaging design will be rolled out across our current product range. It’s a very exciting time to be involved with Almus and Alliance Healthcare.”

Product information
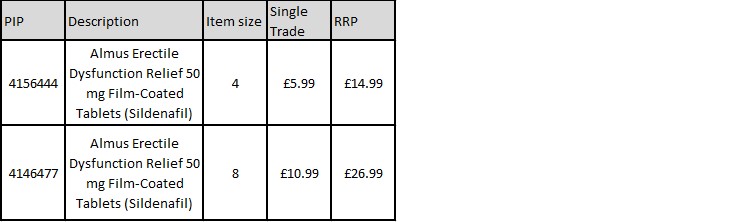
Almus Erectile Dysfunction Relief 50mg Film Coated Tablets are available in both 4 and 8 pack sizes. See pricing and PIP code detail below. Order Almus products through the Alliance Healthcare Portal, AH Direct, your PMR or call: 0330 100 0448
Almus Erectile Dysfunction Relief Sildenafil ‘P’ Medicine PI. Essential Information Name of Product Almus Erectile Dysfunction Relief 50 mg Film-Coated Tablets. Active Ingredient: Sildenafil (as citrate). Indications Almus Erectile Dysfunction Relief 50 mg Film-Coated Tablets is indicated in adult men with erectile dysfunction, which is the inability to achieve or maintain a penile erection sufficient for satisfactory sexual performance. For Almus Erectile Dysfunction Relief 50 mg Film-Coated Tablets to be effective, sexual stimulation is required. Dosage: The recommended dose is one 50 mg tablet taken with water approximately one hour before sexual activity. The maximum recommended dosing frequency is once per day. If Almus Erectile Dysfunction Relief 50mg Film-Coated Tablets is taken with food, the onset of activity may be delayed compared to the fasted state. Legal Category: P Pharmacy OTC status. MANumber PL 08553/0621. MA Holder: Dr. Reddy’s Laboratories (UK) Ltd, 6 Riverview Road, East Yorkshire, HU17 0LD. Further information about this product including adverse reactions, precautions, contraindications and methods of use can be found at: http://www.medicines.org.uk/emc/product/11008/smpc Adverse events should be reported. Reporting forms and information can be found at: http://www.mhra.gov.uk/yellowcard. Adverse events may also be reported to Pharmacovigilance at Dr Reddy’s Laboratories via drreddysgb@eu.propharmagroup.com. Please consult the summary of product characteristics before recommending.
*https://www.baus.org.uk/patients/conditions/3/erectile_dysfunction_impotence

 Spain
Spain